Fight pressure with pressure

Avance® Solo Adapt is indicated for use on pressure ulcers, where it is desirable/necessary to be able to customise the dressing to fit to contoured anatomy in order to achieve a seal.
Parts and features of the system

1. Controlled Fluid Management (CFM) technology
Controlled Fluid Management (CFM) technology is a combination of controlled air flow, absorptive dressing and distal canister, enabling the Avance Solo NPWT system to deliver continuous regulated negative pressure 1 2 3 .

2. Pump
Regulates pressure to deliver a continuous -125mmHg negative pressure 4 . Regular cycling of the pump ensures that fluid is constantly transported to the canister and that the vacuum is maintaned at target levels 4 5 6 .
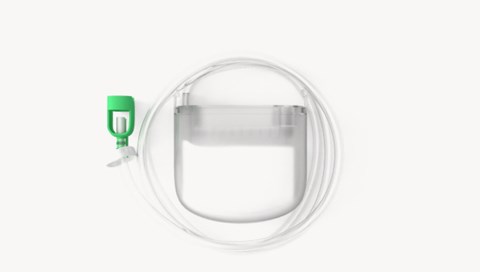
3. 50 ml canister
Excess fluid is transferred to the canister, reducing potential for the dressing becoming fully saturated 2 . The canister may be replaced by clinician, lay carer or the patient themselves 7 8 .

4. Safety
Visual signals and audible alarms for leakage, blockage and low battery help to ensure that loss of therapy or system issues are promptly addressed to enable therapy to continue 7 8 4 9 .
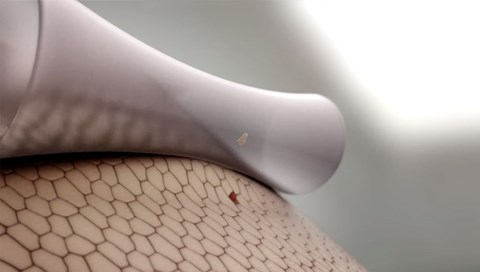
5. Safetac Technology
Avance Solo Border dressings Safetac® adhesive has been proven to minimise trauma to wound site and surrounding skin upon dressing removal 10 11 12 13 , and to minimise pain to the patient during dressing removal 14 15 .

6. Avance Solo Adapt Film and Foam
A conformable film that produces an effective seal for negative pressure therapy 8 2 , as well as a 3 cm foam that can be cut to the shape of the wound and fill the wound cavity. Can be used in conjunction with Mepitel® wound contact layer, to avoid potential for foam to stick to tissue.
Avance solo
How to applyWatch this instructional video to learn how to apply the Avance Solo Adapt system.
using the systems
How it worksOur guides on how to use Avance Solo Adapt includes things such as how to change the canister, achieving a seal, as well as what the various alarms mean and other smart tips and tricks. Click below to learn more.
HOW CAN AVANCE SOLO MAKE A DIFFERENCE TO YOUR PATIENTS, TEAM, AND BUDGET?
'References'
- Data on file (ref. 18)
- Data on file (ref 10).
- Data on file (ref 23).
- Data on file (ref 15)
- Data on file (ref 9)
- Data on file (ref 17).
- Data on file (ref. 13)
- Data on file (ref 9).
- Data on file (ref. 16)
- Van Overschelde P, et al. A randomised controlled trial comparing two wound dressings used after elective hip and knee arthroplasty. Poster presentation at the 5th Congress of WUWHS, Florence, Italy, 25-29 Sep, 2016.
- Silverstein P, et al. An open, parallel, randomized, comparative, multicenter study to evaluate the cost-effectiveness, performance, tolerance, and safety of a silver-containing soft silicone foam dressing (intervention) vs silver sulfadiazine cream. J Burn Care Res. 2011;32(6):617-626.
- Gee Kee EL, et al. Randomized controlled trial of three burns dressings for partial thickness burns in children. Burns. 2015;41(5):946-955.
- David F. et al. A randomised, controlled, non-inferiority trial comparing the performance of a soft silicone-coated wound contact layer (Mepitel One) with a lipidocolloid wound contact layer (UrgoTul) in the treatment of acute wounds. International Wound Journal, 2018.
- Patton ML, et al. An open, prospective, randomized pilot investigation evaluating pain with the use of a soft silicone wound contact layer vs bridal veil and staples on split thickness skin grafts as a primary dressing. J Burn Care Res. 2013;34(6):674-681.
- Bredow J, et al. Randomized clinical trial to evaluate the performance of a flexible self-adherent absorbent dressing coated with a soft silicone layer after hip, knee or spinal surgery in comparison to standard wound dressing. Poster presentation at the 5th Congress of WUWHS, Florence, Italy, 25-29 Sep 2016.




